Lead Chromate Chrome yellow
What is chrome yellow ?
Detailed description
| Chinese name | 铬黄 |
| English name | Lead chromate / Chrome yellow / Chromium yellow |
| Chemical formula | PbCrO4 |
| Molecular weight | 323.18 |
| CAS registration number | 7758-97-6 |
| EINECS registration number | 231-846-0 |
| Exterior | Yellow monoclinic crystal |
| Density (g/mL 25ºC) | 6.12 |
| Melting point (ºC) | 844 |
| Solubility(mg/mL) | Soluble in alkali and inorganic acids, insoluble in water and insoluble in oils |
Properties and Stability
1. It will not decompose if used and stored according to specifications.
2. Avoid contact with oxides.
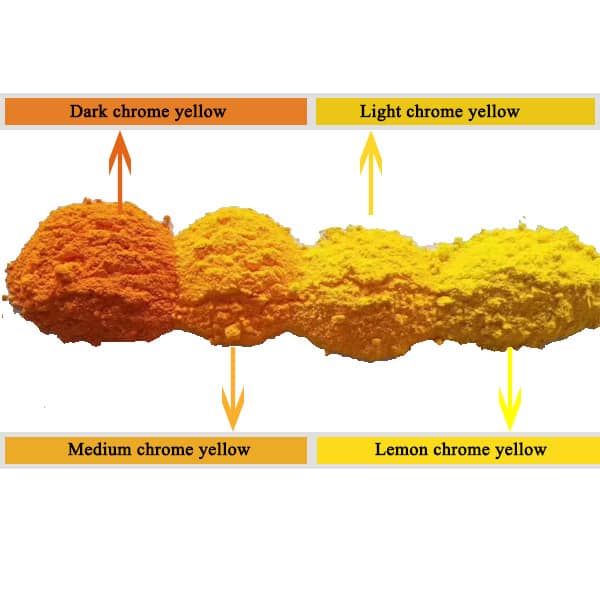
3. It has high tinting power and strong covering power, and will not pulverize in the atmosphere. The color will darken after being exposed to the sun for a long time, and it will easily turn black when exposed to hydrogen sulfide gas. The color light varies with the raw material ratio and manufacturing conditions. The products include lemon chrome yellow, light chrome yellow, medium chrome yellow, dark chrome yellow and orange chrome yellow. poisonous!
Storage method
2. Place in a tight container and store in a cool, dry place.
Uses of chrome yellow
1. Used as raw material for oily and synthetic resin coatings, printing inks, watercolors, oil colors, and pigments. It is also used as a coloring agent for colored paper, rubber and plastic products. It is widely used in industries such as coatings, rubber, plastics, ceramics and pigments.
2. Mainly used in coatings, plastics and inks that require high light resistance, as a yellow pigment with suitable price and performance.
3. Used as analytical reagents. It can also be used as yellow pigment, oxidant, etc.
Differences from other pigments
PbCrO4 is neutral lead chromate. It is a very beautiful and slightly warm color. It is toxic and has certain carcinogenicity.
Lead chromate produced according to different production methods can range from the lightest lemon yellow, light yellow, medium yellow, dark lead chromate to orange. The PbCrO4 sold on the market are all filled with chromium color, such as medium chrome yellow.
PbCrO4 has strong covering power, is opaque and dries quickly. Its stability changes significantly with the depth of its color. The darker the color, the more durable it is. Light chrome yellow will darken when heated or aged, so oil paintings such as Van Gogh’s “Sunflowers” The chromium yellow in the work is far from magical at first, but bright and dazzling.
The stability of lead chromate yellow and common blue will be improved after mixing, but chrome yellow cannot be mixed with sulfur colors, and it will easily become dark and black after mixing. PbCrO4 cannot be mixed with lead white. Lead chromate is rarely seen on the market this century.
The chemical composition of PbCrO4 is chromium sulfide. Chromium sulfide itself is highly toxic, but organic coating technology can greatly reduce contact toxicity.
The medium butter painting color is PbCrO4, which has good light resistance, does not discolor for a long time, is very stable, and dries slowly.
In addition to medium yellow, the light yellow, dark yellow, and orange colors in oil paintings are also cadmium colors or mixtures of cadmium sulfide and other pigments. These are very stable colors. There is also a kind of cadmium tartrazine, which is much more stable than tartrazine, but has poor tinting power. It turns orange when exposed to direct sunlight.
PbCrO4 has strong covering power and can be blended with almost all pigments. It reacts to uncommon copper pigments and easily turns black after blending. When blended with chromium green, the color will deepen after drying.
Danger
Hazard summary: This product is non-flammable, carcinogenic and irritating.
Routes of entry: Inhalation, ingestion.
Health hazards: Acute poisoning: It is irritating to the upper respiratory tract after inhalation; after ingestion, it can cause dizziness, headache, nausea, vomiting, gastrointestinal tract irritation, and can be fatal.
Chronic effects: Can cause anemia, kidney damage, lead accumulation, and lead poisoning. Can cause dermatitis and eczema. Chromium compounds can cause chromium rhinorrhea and skin ulcers. The International Center for Research on Cancer (IARC) lists “chromium and certain chromium compounds” as chemicals that are carcinogenic to humans.
First-aid
1. Skin contact: Take off contaminated clothing and rinse with plenty of running water.
2. Eye contact: Lift your eyelids and rinse with running water or saline. Seek medical attention.
3. Inhalation: Leave the site to fresh air. If breathing is difficult, give oxygen. Seek medical attention.
4. Ingestion: Gastric lavage. Give milk or egg white to drink. Seek medical attention.
Leakage treatment
Isolate the leaked contaminated area and restrict access. It is recommended that emergency responders wear dust masks (full face masks) and protective clothing. Avoid raising dust, sweep it up carefully, put it in a bag and transfer it to a safe place. If there is a large amount of leakage, cover it with plastic sheeting or canvas. Collect and recycle or transport to waste disposal site for disposal.
Operational disposal
Precautions for handling and handling: Close operation and enhance ventilation. Operators must undergo special training and strictly abide by operating procedures.
It is recommended that the operator wear a dust mask (full face mask), a one-piece tape respirator suit, and rubber gloves. Keep away from flammable and combustible materials. Avoid dust generation. When handling, load and unload with care to prevent damage to packaging and containers. Equipped with leakage emergency treatment equipment. Empty containers may be harmful residues.
Disposal
Disposal method: Dispose in accordance with relevant national and local regulations. Or contact the vendor or manufacturer to determine disposal methods.
Control protection
1. Respiratory system protection: When you may come into contact with its dust, you must wear a dust mask (full face mask). During emergency rescue or evacuation, air respirators should be worn.
2. Eye protection: Protection has been included in respiratory protection.
3. Body protection: Wear a one-piece tape-type anti-virus suit.
4. Hand protection: Wear rubber gloves.
5. Other protection: Maintain good hygiene habits. Conduct pre-employment and regular physical examinations